Background
Dry eye disease (DED) is one of the most common presentations in eye care offices.1 The symptoms range from intermittent computer-related discomfort to severe debilitating dryness. The Tear Film & Ocular Surface Society (TFOS) defined dry eye disease as “a multifactorial disease of the ocular surface characterized by a loss of homeostasis of the tear film and accompanied by ocular symptoms, in which tear film instability and hyperosmolarity, ocular surface inflammation and damage, and neurosensory abnormalities play etiological roles.”2
It has been demonstrated that signs and symptoms of DED do not correlate.3 Some patients have significant symptoms with few observable signs. In contrast, others demonstrate severe ocular surface disease with minimal symptoms. Although the degree of DED can be placed on a continuum, it is essential to identify patients with Sjögren’s syndrome (SS)-related DED, as eye care practitioners are often the starting point of a complete diagnosis for these patients.
Case Presentation
History
A 32-year-old female presented with symptoms of reduced contact lens comfort and wearing time, scratchy, burning eyes that worsened with prolonged computer use, and photosensitivity. She wondered if she should have LASIK to correct her vision. She stated that she had always had good health and did not take any medications. She found that using ocular lubricants helped when her eyes felt particularly irritated.
Examination
Best corrected visual acuities were 20/20 in both eyes. Binocular functions were normal. Refraction was -3.50 -0.25×090 in the right eye (OD) and -4.25 -0.50×090 in the left eye (OS). Intraocular pressures were OD 11 mmHg and OS 13 mmHg.
Biomicroscopy revealed a debris-filled tear film. The conjunctivas and lid margins were mildly hyperemic. The corneas appeared clear in both eyes under white light examination.
The fundus examination was within normal limits.
Dry Eye Examination
Schirmer scores were OD 5 mm and OS 7 mm in 5 minutes.
Tear break-up time with sodium fluorescein (NaFl) was OD 5 seconds and OS 3 seconds.
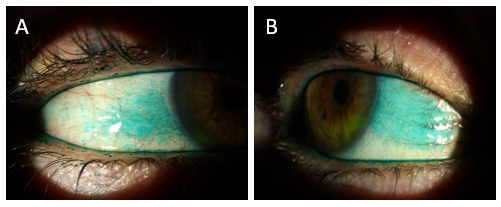
Corneal staining was graded as OD Grade 2 and OS Grade 1 out of a maximum of 3 at 5 minutes post NaFl insertion (Figure 1).
 Figure 1: Corneal staining 1 minute (A) and 5 minutes (B) after instillation of sodium fluorescein
Figure 1: Corneal staining 1 minute (A) and 5 minutes (B) after instillation of sodium fluorescein
Lissamine green staining was Grade 3 out of a maximum of 3 nasal and temporal OU (Figure 2).
Figure 2: Lissamine green staining of the temporal (A) and nasal (B) conjunctiva
There was mild meibomian gland dysfunction and little dropout was visible with meibography.
Additional History
Upon questioning, the patient reported that her mother suffered from scleroderma and her maternal aunt suffered from rheumatoid arthritis. She admitted to a dry mouth and recent cavities on her last visit to the dentist. She did not believe that she could eat five crackers without drinking water. She had noticed a dry vagina and recent recurrent yeast infections. She also recognized that she was experiencing increased fatigue and mild arthralgia (joint pain).
Diagnosis and Treatment Plan
The patient was diagnosed with aqueous deficient and evaporative dry eye that was likely related to SS. A lengthy discussion ensued, with explanations of the nature of this autoimmune disease and the inflammatory mechanism that was interfering with the lacrimal gland’s ability to secrete fluid and the subsequent inflammation present on her ocular surfaces. It was explained to the patient that this condition was chronic. However, she was reassured that it rarely causes permanent vision loss and that she should be able to manage her eyes with routine treatment and regular examinations.
Treatment Plan
A report was sent to the patient’s family doctor with a recommendation for a full autoimmune blood workup, including anti-Ro, anti-La and antinuclear antibody (ANA). The patient was placed on loteprednol etabonate ophthalmic suspension (Lotemax) four times per day (QID) for one month and topical cyclosporine 0.05% twice per day (BID) ongoing. She was introduced to hyaluronate-based preservative-free lubricants, to be used in place of the loteprednol suspension, following completion of the four-week course. No contact lens wear was allowed.
One Month Assessment
The patient returned to the office after one month. She reported that she had followed the drop regime as prescribed. She noted some stinging on insertion with the topical cyclosporine but that it had diminished over the month. Her eyes felt “much better” and she noticed less sensitivity to light.
The report from her family doctor confirmed anti-Ro and ANA positive testing and said that she had been referred to a rheumatologist for a confirmation of SS.
The dry eye examination demonstrated minimal change in the conjunctivas but some reduced fluorescein staining of the corneas.
The issue of LASIK was raised and the patient was advised that this was not recommended because of the increased dry eye problems that can occur after this procedure. She expressed a desire to go back to wearing her contact lenses and the risk of this behaviour was discussed. It was agreed that she would reduce the number of days and hours of wear and that she would be refit with daily disposable lenses. Lubricating drops were to be used QID when wearing her contact lenses.
Discussion
DED is a common presentation in eye care offices. The prevalence increases with age and female sex.4 Rheumatic auto-immune diseases are often associated with DED, with SS being the most likely. SS is one of the rheumatic auto-immune diseases that includes rheumatoid arthritis, scleroderma, and systemic lupus erythematosus. SS affects 0.1% to 4.8% of the population, with a female-to-male ratio of 9:1, depending on the cohort studied, classification criteria and methodology used.5 The age of onset is variable but is generally stated as from 40 to 60 years of age. However, young women and men can also be affected. Patients can spend many years going from doctor to doctor before having a confirmed diagnosis.6 A diagnosis is essential for many reasons, including the many complications that can occur in SS, including lymphoma.7
Optometrists have an essential role in all patients exhibiting DED, particularly in identifying those patients with SS. Taking a careful history is an important part of the examination. The simple questions of dry mouth, arthralgia, fatigue, and family history of autoimmune disease can lead the examiner down the right path. The diagnosis requires more than the eye examination, as blood work is a necessary step. If the blood work is negative, a referral to an ear, nose and throat (ENT) doctor is required for a lip biopsy. Diagnosis requires positive blood work or lip biopsy and at least one eye sign of Schirmer tests of ≤5 mm in one eye OR staining of at least 4 out of 9 in one eye. The American College of Rheumatology (ACR)/European League Against Rheumatism (EULAR) classification criteria requires a total score of ≥4, based on the criteria8 shown in Table 1.
| Item | Score |
| Labial salivary gland with focal lymphocytic sialadenitis and focus score of ≥1 foci/4 mm2 | 3 |
| Anti-SSA/Anti-Ro positive | 3 |
| Ocular staining score ≥5 (or van Bijsterveld score ≥4) in at least one eye | 1 |
| Schirmer’s test ≤5 mm/5 minutes in at least one eye | 1 |
| Unstimulated whole saliva flow rate ≤0.1 ml/minute | 1 |
Ongoing management of these patients surrounds monitoring their symptoms and signs. Routine ocular surface assessments at six-month intervals are recommended. Intermittent use of topical steroids is often required, with appropriate monitoring. Additional options include autologous serum drops or platelet-rich plasma (PRP) drops for severe cases.9,10
There is some evidence that lifestyle modifications can be helpful for these patients. Humidifying the office and bedroom,11 the use of corrective glasses or goggles at the computer,12 drinking plenty of water,13 eating fresh, non-processed foods to reduce general inflammation,14 getting plenty of rest,15 and avoidance of air currents16 can improve symptoms and signs.
Clinical Pearl
Always entertain the possibility of SS in women presenting with DED. Simple questions about dry mouth and fatigue can improve the likelihood of that diagnosis. Corneal staining is important, but the conjunctiva is most often affected by SS. If not widely available, lissamine green can be compounded at a pharmacy and is worth using for this reason. Corneal staining should be viewed after 3 to 5 minutes to ensure that the grading is accurate.17 There are only so many tools in our DED toolbox, and unfortunately, no specific medication is available for SS patients. However, educating the patient about their disease and routinely observing them will help them in the management of their particular type of DED.
REFERENCES
- Stapleton F, Alves M, Bunya V, et al. TFOS DEWS II epidemiology report. Ocul Surf. 2017;15(3):334-68.
- Craig JP, Nichols KK, Akpek EK, et al. TFOS DEWS II definition and classification report. Ocul Surf. 2017;15(3):276-83.
- Nichols K, Nichols J, Mitchell G. The lack of association between signs and symptoms in patients with dry eye disease. Cornea. 2004;23(8):762-70.
- Schaumberg DA, Sullivan DA, Dana MR. Epidemiology of dry eye syndrome. Adv Exp Med Biol. 2002;506 (PtB):989-98.
- Binard A, Devauchelle-Pensec V, Fautrel B, et al. Epidemiology of Sjögren’s syndrome: Where are we now? Clin Exp Rheumatol. 2007;25(1):1-4.
- Huang Y-H, Lu T-H, Chou P-L, Weng M-Y. Diagnostic delay in patients with primary Sjögren’s syndrome: A population-based cohort study in Taiwan. Healthcare. 2021;9(3):363-70.
- Sikara M, Ziogas D, Argyropoulou P, et al. Ten-year overall survival and standarized mortality ratio in the largest single center cohort of patients with primary Sjogren’s associated lymphomas. Ann Rheum Dis. 2019;78:1179.
- Shiboski C, Shiboski S, Seror R, et al. 2016 American College of Rheumatology/European League Against Rheumatism classification criteria for primary Sjögren’s syndrome: A consensus and data-driven methodology involving three international patient cohorts. Arthritis Rheumatol. 2017;69(1):35-45.
- Wróbel-Dudzińska D, Przekora A, Kazimierczak P, et al. The comparison between the composition of 100% autologous serum and 100% platelet-rich plasma eye drops and their impact on the treatment effectiveness of dry eye disease in primary Sjogren syndrome. J Clin Med. 2023;12(9):3126.
- Cui D, Li G, Akpek E. Autologous serum eye drops for ocular surface disorders. Curr Opin Allergy Clin Immunol. 2021;21(5):493-9.
- Mandell JT, Idarraga M, Kumar N, Galor A. Impact of air pollution and weather on dry eye. J Clin Med. 2020;9(11):3740.
- Korb DR, Blackie CA. Using goggles to increase periocular humidity and reduce dry eye symptoms. Eye Contact Lens. 2013;39(4):271-6.
- Walsh NP, Fortes MB, Raymond-Barker P, et al. Is whole-body hydration an important consideration in dry eye? Invest Ophthalmol Vis Sci. 2012;53(10):6622-7.
- Wu Y, Xie Y, Yuan Y, et al. The Mediterranean diet and age-related eye diseases: A systematic review. Nutrients. 2023;15(9):2043.
- Yu X, Guo H, Liu X, et al. Dry eye and sleep quality: A large community-based study in Hangzhou. Sleep. 2019;42(11):1-8.
- Yu Z, Yazdanpanah G, Alam J, et al. Induction of innate inflammatory pathways in the corneal epithelium in the desiccating stress dry eye model. Invest Ophthalmol Vis Sci. 2023;64(4):8.
- Begley C, Caffery B, Nelson J, Situ P. The effect of time on grading corneal fluorescein and conjunctival lissamine green staining. Ocul Surf. 2022;25:65-70.
Get the latest Optical News delivered to you. Click here to subscribe to our FREE print magazine and weekly newsletter